
Leave message
Can’t find what you’re looking for?
Fill out this form to inquire about our custom protein services!
Inquire about our Custom Services >>

































 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free! Get your ComboX free sample to test now!
Get your ComboX free sample to test now!
 Time Limited Offer: Welcome Gift for New Customers !
Time Limited Offer: Welcome Gift for New Customers !  Shipping Price Reduction for EU Regions
Shipping Price Reduction for EU Regions
Antibody test, also known as the serology test, is an important supplementary diagnosis of COVID-19. In addition to the nucleic acid test, the antibody test can help with not only the accuracy of diagnosis but also the characterization of the prevalence and spread of the disease. Antibody test will identify if a person has been exposed to SARS-CoV-2 and developed immunity against SARS-CoV-2 infection, which is better suited for public health surveillance and vaccine development.
So far, the FDA has issued the Emergency Authorization Use to 12 antibody tests, including 3 colloidal gold assays, 4 chemiluminescent assays, and 5 ELISA assays (Table 1). Due to urgency and demand, a lot of antibody tests are rapidly developed and made available on the market with limited validation. To address the problem, some scientists are evaluating these assays using clinical samples. In this article, we will take a close look at the performance of the ELISA assays. (Find more about the performance of colloidal gold and chemiluminescence methods.)
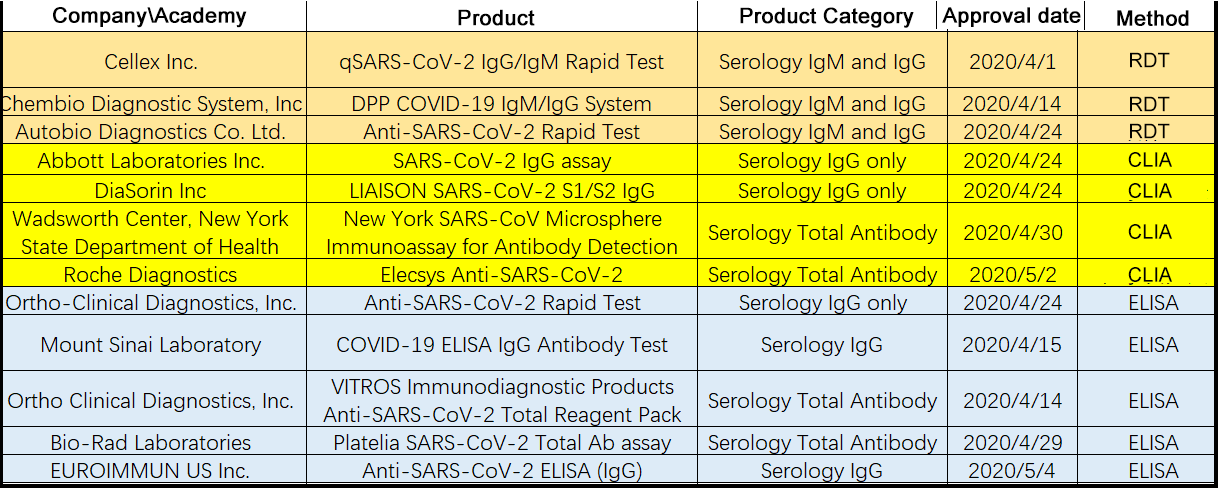
Table 1. Emergency Use Authorizations for COVID-19 by FDA
Beijing Wantai Biological Pharmacy Enterprise Co., Ltd developed 3 enzyme-linked immunosorbent assay (ELISA) kits against the total antibodies (Ab), IgM, and IgG of SARS-CoV-2 respectively. Briefly, the ELISA for Ab was developed based on double-antigens sandwich ELISA, using the mammalian cell-expressed recombinant receptor binding domain (RBD) of the spike protein of SARS-CoV-2 and HRP-conjugated antigen. The IgM μ-chain capture ELISA was used to detect the IgM antibodies using the same HRP-conjugated antigen. The IgG antibodies were measured using an indirect ELISA kit based on a recombinant nucleocapsid protein.
173 patients were enrolled in this study. The seroconversion rate for Ab, IgM, and IgG was 93.1% (161/173), 82.7% (143/173), and 64.7% (112/173), respectively. Twelve patients who remained seronegative for Ab testing possibly due to that their samples involved were all collected at the early stage of illness. The specificity of the assays for Ab, IgM, and IgG was determined as 99.1% (211/213), 98.6% (210/213), and 99.0% (195/197) by using the samples collected from healthy individuals.
The performances of the different ELISA tests and the nucleic acid test in samples with different duration of illness were also analyzed. As the duration of illness increased, the sensitivity of ELISA tests kept increasing and surpassed that of the nucleic acid test after day 8.
Above all, combined use of the nucleic acid test and ELISA test is believed to improve markedly the sensitivities of COVID-19 diagnosis in different stages.
Another group from Denmark also investigated the performance of 3 commercially available ELISA kits, including a Wantai Ab ELISA kit we just mentioned and two ELISA kits from Euroimmun (IgG and IgA respectively), which gained EUA from FDA in May (Table 1). The IgA or IgG antibodies against SARS-CoV-2 spike protein subunit 1 (S1) are detected by using an indirect ELISA format.
As shown in the ELISA data distribution figure (Figure 1), the positive and negative data points were distinct for the Wantai total Ab assay, with a cut-off value above all the control serum samples, which allowed for unequivocal interpretation. Conversely, the Euroimmun IgA and IgG assays data had a less distinct separation.
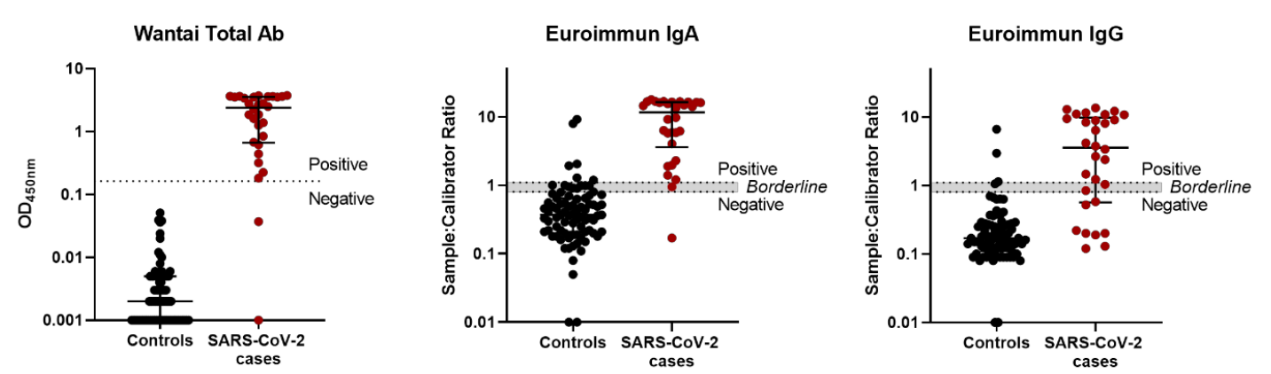
Figure 1. SARS-CoV-2 antibody ELISA assay performance
Overall, the Wantai total Ab ELISA had superior sensitivity and specificity compared to both Euroimmun IgA and IgG ELISAs (Table 2). This notably lower sensitivity for SARS-CoV-2-specific IgG detection is in agreement to that observed in the previous study we talked about (Wantai IgG ELISA kit). The possibility that overall lower sensitivity of SARS-CoV-2 IgG ELISAs may be a more universal occurrence rather than manufacturer dependent warrants further investigation.
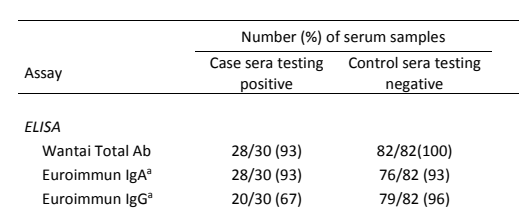
Table 2. Analytical sensitivities and specificities for ELISA kits
In addition to lower sensitivities, the Euroimmun IgA and IgG ELISAs are also more prone to cross-react with negative serum samples. The difference performances between the assays may be partially explained by the ELISA format and the antigens they used. The nucleocapsid and spike proteins are critical reagents for the antibody detection kits, which could greatly affect the performance of the assay.
ACROBiosystems has developed S1, N, wild type RBD as well as mutant RBD proteins with various tags including His, Fc, mFc and His & Avi, which are suitable for COVID-19 serological test development. These proteins can be applied to a colloidal gold method, chemiluminescence method, and ELISA method. ACRO is gaining lots of positive feedback for the high-quality SARS-CoV-2 antigen proteins.
Product:SARS-CoV-2 (COVID-19) Nucleocapsid protein, His Tag
The sensitivity of N protein binding to Anti-N mAb is 0.02 ng/mL as verified by ELISA.
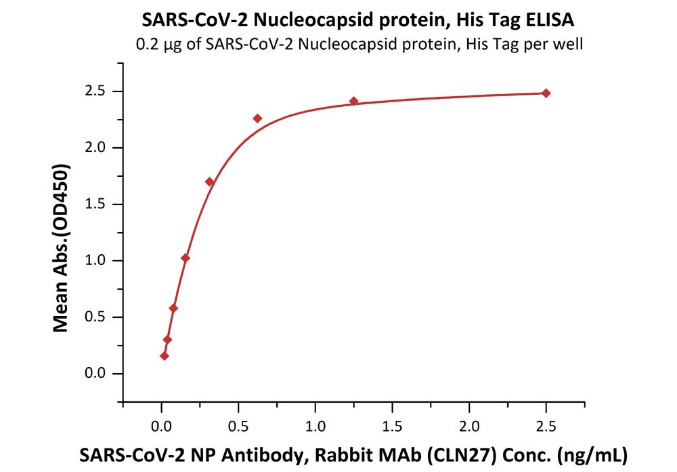
Immobilized SARS-CoV-2 Nucleocapsid protein, His Tag (Cat. No. NUN-C51H9) at 2 μg/mL (100 μL/well) can bind SARS-CoV-2 NP Antibody, Rabbit MAb (CLN27) with a linear range of 0.02-0.3 ng/mL.
Product:SARS-CoV-2 (COVID-19) S protein RBD, His Tag (MALS verified)
The sensitivity of RBD protein binding to ACE2 protein is 0.008 ng/mL as verified by ELISA.
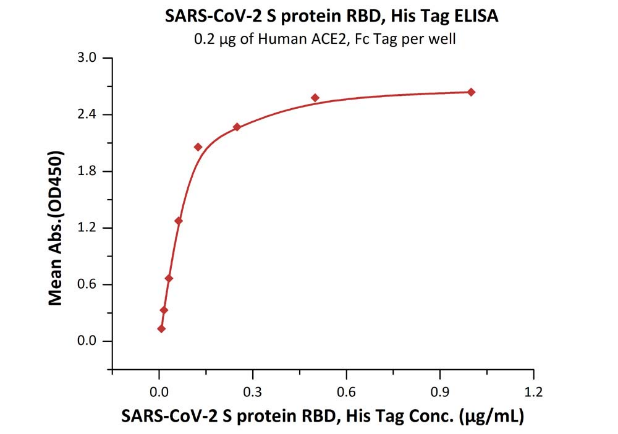
Immobilized Human ACE2, Fc Tag (Cat. No. AC2-H5257) at 2 μg/mL (100 μL/well) can bind SARS-CoV-2 S protein RBD, His Tag (Cat. No. SPD-C52H3) with a linear range of 0.008-0.125 μg/mL.
Reference:
1. Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019
2. Evaluation of nine commercial SARS-CoV-2 immunoassays
This web search service is supported by Google Inc.







